Dr. Gross has now performed over 2500 Hip Surface Replacement (HSR) procedures over the last 12 years. Most failures occur during the first 6 months of the healing period. However, there is a slow rate of failure that occurs over time. Therefore the overall failure rate increases for a group of patients as the length of follow-up increases. In our recent publication in the Journal of Arthroplasty 2011, we reported that our Corin Hybrid HSR achieved a 93% survivorship at 11 years follow-up. Longer-term data is not available. Multiple improvements have been made since this initial patient group. Our most recent cases use the Biomet uncemented Recap /Magnum. We report here the early results of the first 1000 done between March 2007 and July 2010 with a 99.4% rate of follow-up (90% completely up to date on their follow-up, and 64 % achieving at least 2 years follow-up). Not all complications lead to failure. Below is a complete list of major complications (not just failures) in the first 1000 uncemented HSR using the Biomet system:
A.) Failures Requiring Revision Surgery (1000 cases):
1. Femoral neck fracture: 6
2. Early femoral collapse (avascular necrosis): 2
3. Failure of acetabular ingrowth: 5
4. Adverse wear failure: 2
5. Deep infection with loss of implant: 0
6. Recurrent dislocations requiring revision: 0
7. Femoral component loosening: 0
8. Acetabular component loosening: 0
9. Subtrochanteric femur fracture (related to hardware removal): 1
TOTAL: 16
B.) Cases requiring significant repeat surgery (1000 cases):
1. Traumatic intertrochanteric fracture (5 and 11 months postop): 2
2. Deep infection (cured): 2
3. Significant superficial infection (cured): 1
4. Frostbite from ice machine: 2
TOTAL: 7
C) Other Complications (1000 cases):
1. Dislocations: 2
2. Pulmonary emboli: 3
3. Deep vein thrombosis: 2
4. GI bleed requiring transfusion: 1
5. Minor stroke: 1
6. Nerve injuries: 0
7. Postoperative transfusions: 0
8. Femoral notches: 0
9. Vascular injuries: 0
10 Deaths: 0
TOTAL: 9
Please see survivorship curves on the back of this page.
Past results do not guarantee future complication rates. Although the above represent the most common complications associated with this procedure, others could also occur. We continue to strive to make improvements, and hope that these complication rates can be further decreased as we gain even more experience.
- Dr. Gross is the operating surgeon (No trainee will perform your operation).
- Dr. Gross developed the Biomet implants but no longer receives royalties for these implants.
- Biomet Recap and Magnum components are FDA approved. Use as a total hip resurfacing is however considered off-label.
- Information from your treatment is used for research purposes, but you will not be identified.
If you have any questions about the above information, please don’t hesitate to ask
I have reviewed the above and understand the risks involved with this operation. I would like Dr. Thomas Gross to perform Hip Surface Replacement on me.
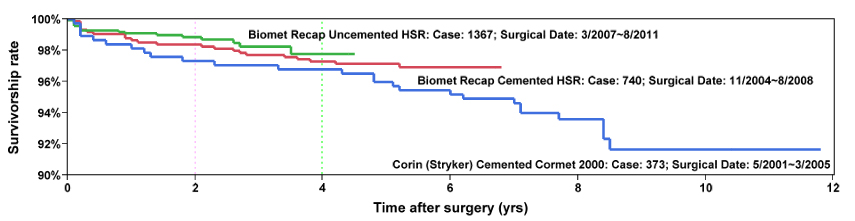
HIP RESURFACING SURVIVORSHIP
Survivorship of hip resurfacing continues to improve as we gain more experience and find measures to prevent failures. Theses survivorship curves give the reader an opportunity to see what the odds are that their implant will still be functioning at some time point after implantation. We have used three implant systems in the last 10 years. The first were hybrid fixation Corin devices (blue), the second were hybrid fixation Biomet devices (red), the latest are completely uncemented Biomet devices (green).
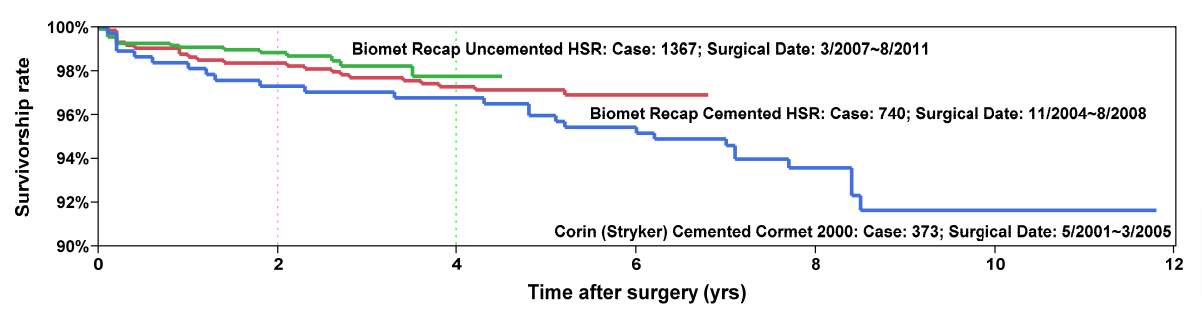
In the last 10 years we have learned what patients with particular characteristics are at higher risk for failure. The strategy of many experts is patient selection. If they avoid performing hip resurfacing on higher risk patients, their overall results will improve. I disagree with this defeatist strategy.
My improving results are not achieved by patient selection. I have always practiced minimal patient selection and my criteria have not changed much over the last 10 years. Instead, my goal is to find treatment modifications to improve the results in patients that are traditionally identified as higher risk. In some cases, we have already accumulated scientific evidence of improved results; in other cases, we still need more time to gather data to be certain that our treatment modifications have improved results. Examples of our innovations include:
- Femoral neck fracture: stratifying risk of femoral neck fracture by bone density and BMI and treating higher risk patients with slower weight bearing and bisphosphonate drugs. We have demonstrated that this substantially reduces risk.
- Failure of acetabular implant attachment: dysplasia patients are at higher risk because of socket deformities. Use of Trispike acetabular components in severely deficient sockets has eliminated these failures in this high‐risk group.
- Femoral cysts: Bone grafting cysts instead of filling them with cement has resulted in eliminating femoral cysts as a risk factor for failure in our patients.
- Femoral Loosening: The major source of late failure in my cemented femoral components. We have demonstrated that uncemented femoral components are equally as good as cemented ones at up to four years follow‐up. Our hypothesis is that uncemented femoral components will be less likely to loosen in the long term.
- Adverse wear failure: This has been linked to acetabular component position, particularly high inclination angles (AIA). We have developed an intraoperative XR technique that has lowered the chance of implanting a component with an AIA>50 from 26% to 4%. Recent improvements in our technique are focused on still further improvements in the odds of achieving ideal component position.
I have been performing hip resurfacing for over 10 years and have maintained an accurate database of current patients with an overall >90% rate of follow‐up. This chart presents the results for 2480 cases.