Introduction
Metal-on-metal bearings in total hip replacement have experienced a resurgence of popularity in recent times. This is due mainly to a significant number of long-term clinical studies highlighting the remarkably low wear rates of metal-on-metal bearings implanted over the past 40 years.1-4 Clinical complications with the early generation components usually were related to manufacturing issues such as, improper tolerances and poor design. 5 Recent advancements in manufacturing technology have enabled more precise tolerances, surface finish, and designs that provide an increased range of motion (Figure 1). Documented clinical success with modern devices has led to the FDA granting market approval to some orthopedic companies in the US.6,7

Metal-on-metal is particularly attractive in the total hip not only due to the ultra-low wear exhibited in numerous clinical and simulator studies1–8 (Figure 2), but also due to the flexibility afforded by designs based on the properties of cobalt chromium alloy. This flexibility enables the use of large diameter heads in a variety of acetabular configurations. Larger metal-on-metal femoral heads can provide increased range-of-motion and stability, as well as increased resistance to dislocation. 9-12 The incorporation of larger diameter femoral heads into ceramic and polyethylene bearings presents design challenges due to impingement concerns with ceramics, and increased brittleness with thin polyethylene liners, all leading to an increased risk of mechanical fracture.13,14 Metal-on-metal components do not pose a risk of mechanical fracture. Current metal-on-metal designs allow the use of the larger 38mm heads inacetabular component sizes as small as 46mm.
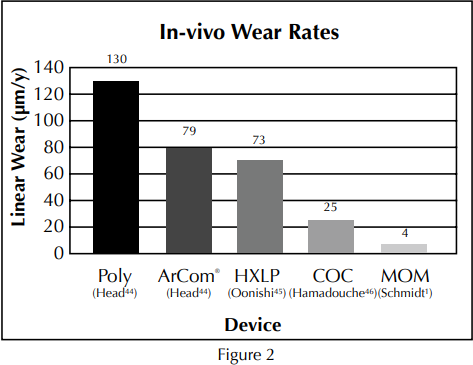
Metal-on-metal has exhibited ultra-low wear rates not only in simulator studies, but also through published in-vivo wear studies as shown in the in-vivo wear comparison above.
Despite its lengthy and positive clinical history, there have been concerns expressed regarding the long-term biological consequences of metal-onmetal wear debris, particularly the release of cobalt and chromium ions into the body. Questions about metal ions and their biological effects are not new. Many studies have been conducted over past decades. All have failed to establish a causal link between ion release, and cancer or other negative systemic effects.15–22 As with each bearing surface, certain unknowns still exist, and in recent times, fueled by rumors and misinformation about the metal ions, the debate lingers on. Following are five reasons why many leading researchers consider the metal ion debate to be inconsequential.
1. Cobalt chrome alloy is a known material.
Vitallium™ or cobalt chromium alloy has been in use in orthopedics for more than 80 years since its first use by Dr. Smith-Petersen in total hip replacement surgery in 1917.23 The strength and durability of cobalt chrome has been applied to a variety of bearing surfaces over the years including total knees, elbows, shoulders and hips. Cobalt chrome was first used in metal-on-metal applications as early as 1958 in designs such as the Ring, Muller, McKee/Farrar, and Sivash prostheses. Since those early days of orthopedics, hundreds of thousands of patients have received metal-on-metal implants, with the majority being implanted in Europe. Interestingly, 40 years and hundreds of thousands of implants later, metal-on-metal ion release remains a non-issue among most European orthopedic surgeons.

Cobalt chrome has been used in a wide variety of orthopedic implants over the past 80 years including this hip implant from the early 20th century.
Throughout the history of metal-on-metal various authors have examined the ion release issue. Following are brief summaries of some of their findings.
The Risk of Cancer Following Total Hip or Knee Arthroplasty, JBJS, May 200115 Dr. Tharani, et al., reported on the risk of cancer following total hip or knee arthroplasty.15 Nine different studies encompassing 140,000 total hip and knee cases were combined and statistically evaluated in order to compare the relative risk of cancer to that of the general population or a control group. In these nine studies, a total of 25 cases of the malignant disease were reported, and 34% of the cancers occurred within the first two years of implantation. These findings showed, “the available data do not support a causal link between total hip arthroplasty and the development of cancer.” In addition, no increased risk of cancer with a metal-onmetal hip implant versus a polyethylene implant was found. Long-term concerns about metal-on-metal in the hip should be weighed against the study data showing that 34% of the cancers after total joint replacement occurred within the first two years and were not associated with the total joint implant.

Cobalt chrome was first used in metal-on-metal applications as early as 1958 in designs such as the Muller, McKee/Farrar, Sivash and Ring (pictured on the left) prostheses.
Cancer Risk After Metal-on-Metal and Polyethylene-on-Metal THA, CORR, August 199616
Dr. Visuri, et al., also studied the cancer risk after metal-on-metal and polyethylene-on-metal total hip arthroplasty.16 The study involved 579 metal-on-metal patients and 1,585 metal-on-polyethylene patients with up to 16-year follow-ups. Their objective was to compare the incidence of cancer after both metal-on-metal and metal-on-polyethylene total hip replacement to that of the general population in Finland. There was no statistically significant increase in cancer of any type, at 16-year follow-up, with either a metal-on-metal or metal-on-polyethylene compared to the expected cancer rate of the general population. The authors concluded that “the observed variation in the incidence of different cancers among patients who had total hip arthroplasty compared with the general population suggests that factors other than total hip arthroplasty play a major role in the origin or cancer” (Figure 3).
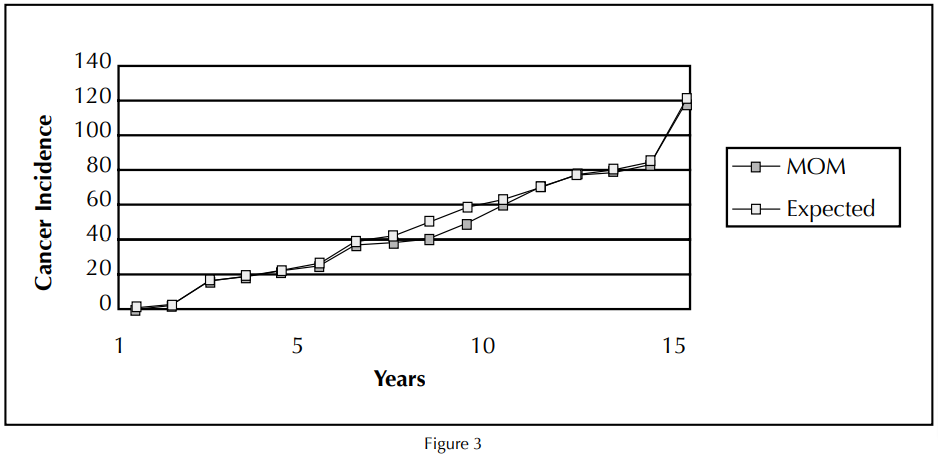
Dr. Visuri reported no statistically significant increase in cancer of any type with either a metal-on-metal or metal-onpolyethylene hip replacement compared to the expected cancer rate of the general population at a 16-year follow-up.
Distribution of Cobalt Chromium Wear and Corrosion Products and Biologic Reactions, CORR, August 1996 17
Dr. Merritt, et al., examined the distribution of cobalt chromium wear and corrosion products and the subsequent biologic reactions.17 The issue of cobalt chromium wear and corrosion products was evaluated in an effort to see how this might relate to the biologic performance of implant devices. The findings were extremely favorable as related to a well functioning metal-on-metal hip implant. The authors conclude, “it is clear at this stage in total joint replacement, that more reported adverse biologic responses are occurring to the polyethylene than to the metal. It is tempting to address this issue by eliminating the polyethylene.”
2. Metal ion release is not unique to metal-onmetal devices.
Metal ions are a byproduct of virtually every metallic implant. Nails, screws, stems, metal cups, plates, modular heads, cages, and cables will each release ions of their perspective metals. This is further evidenced in studies by Dr. Kriebich who demonstrated that loose cobalt chrome stems exhibit equivalent blood ion levels compared to that of a metal-on-metal implant24 (Figure 4)
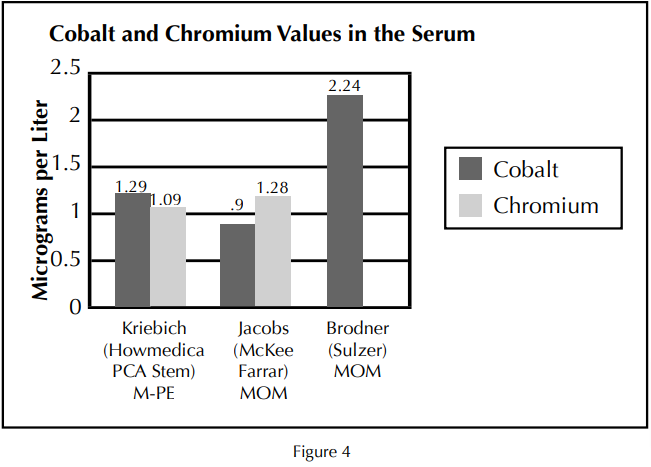
Serum studies of metal ions demonstrate similar values between loose cobalt chrome stems and that of a metal-on-metal implant.

Some ceramic-on-ceramic designs may also release metal ions release due to metal-on-metal neck impingement and fretting at modular taper junctions.
Some ceramic-on-ceramic designs may also release metal ions due to neck impingement and fretting at modular taper junctions.33 There have been case reports of femoral neck notching with some recent ceramic-on-ceramic designs leading to excessive metal debris, resulting in metallosis.25 Clearly, in vivo transfers of metal ions are not unique to metal-on-metal bearings, so the question becomes: if we can’t avoid metal ions in total hip arthroplasty, what is an acceptable amount? Researchers have been trying to define maximum values for cobalt and chromium ions for years. The
main issue they are confronted with is the fact that years of metal ion research have yet to reveal any toxic signifi cancer associated with metal ions released from metal-on-metal articulations.15-22 Therefore, setting a maximum value has not been possible. Data on the effects of cobalt and chromium in the body at levels much higher than that of a metal-on-metal implant can be found in studies performed outside of the fi eld of orthopedics, such as in the metal industry. For general purposes, comparisons can be made to the metal ion levels reported from metal-on-metal implant studies.
3. Case reports and studies of cobalt and chromium in other industries support the use of metal-onmetal bearings in orthopedics.
Workers in the metal industry are typically exposed to higher than-normal levels of the metals they are processing. Tests have shown higher than normal blood ion values in factory workers exposed to chromium and cobalt.
These metals can be introduced into their system through dermal exposure, inhalation and accidental ingestion.27-31 It should be noted that even though these workers demonstrate blood ion values 15-20 times higher than that of a metal-on-metal implant, no systemic toxicity has been established. Metal-onmetal ion levels of chromium can also be compared to “studies on worker cohorts who have historically been exposed well above the current permissible standards (IARC, 1990), and have not shown a consistent pattern of elevated cancer incidence” (Figure 5) (Proctor, 2002).27
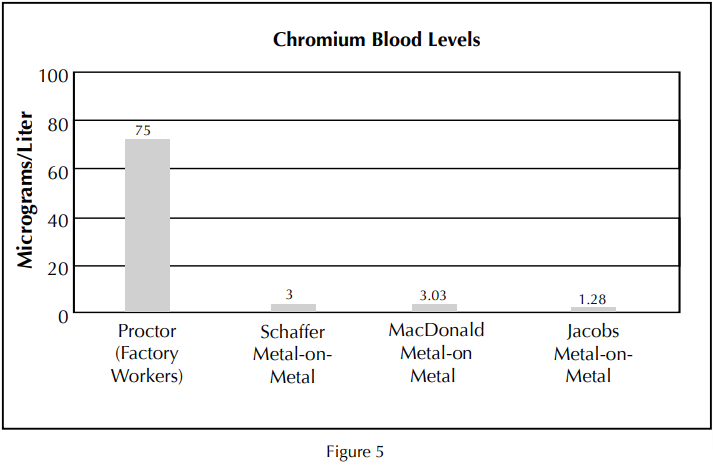
Metal-on-metal ion levels of chromium can also be compared to “studies on worker cohorts who have historically been exposed well above the current permissible standards, and have not shown a consistent pattern of elevated cancer incidence.” 27
For example, discussions of toxicity have also extended to the female population and pregnancy. In the 1960s cobalt was sometimes used as a remedy for anemia in pregnant women. Barceloux reported on one group of women receiving over 100,000 Mcg of cobalt per day, all of whom delivered healthy full-term infants, and experienced no adverse side effects related to the cobalt medication30 (Figure 8).

Dr. Brodner from Vienna, Austria recently reported (Montreal, June 2003) on three pregnant women who were recipients of metal-on-metal hips. Metal ion testing was performed at the time of delivery. Trace amount of ions could be detected in the mothers, with no detectable amount in the umbilical cords. Dr. Brodner concluded that metal-on-metal was safe for women of childbearing age as “the placenta acts as an effective barrier for cobalt and chromium disseminated from metal-on-metal articulations.”31 Not only has cobalt been used as a medicine for refractory anemia, but chromium also has beneficial effects in the body, such as assisting in proper metabolic function.
A debate about metal ion release, similar to that occurring in orthopedics, is currently being waged in the food industry. Some investigators have expressed concern regarding the leaching of metals, such as chromium, into food from metallic cooking utensils. As in the orthopedic field, some have remained cautious, stating that more research in this area is needed. Dr. Aitio, Chief Physician for the Department of Industrial Hygiene and Toxicology at the Finnish Institute of Occupational Health, concluded that: “No toxicity is to be expected from the chromium leached from kitchenware, it may in fact be beneficial to health since the amounts of chromium in present Western-type diets are generally small in comparison to amounts considered to be optimal.”26

Debate about metallic ion release from food utensils.
4. Cobalt and Chromium may be beneficial to the body as established by research and listed by the US government.
At times in the midst of the debates regarding metal ions, it is helpful to remember that metal-on-metal devices are FDA regulated and cleared for orthopedic applications by the US government. The government has also established a recommended daily allowance for chromium. “Chromium III (trivalent chromium) is an essential micronutrient with a recommended daily requirement for humans in the range of 50 to 200 micrograms per day (µg/d).”27 As demonstrated in the chart below, a metalon-metal hip is associated with blood chromium levels of 3 – 5 Mcg/l, the majority of which is excreted though the urine.18
As with many elements, chromium can exist in several atomic valence states. Chromium VI (hexavalent) and Chromium III (trivalent) are the two most common. “Chromium III displays extremely low or no toxicity via all routes of human exposure and does not pose a carcinogenic hazard (ATSDR, 2000; U.S. EPA, 1998a; IARC, 1990; WHO, 1988).”27 Chromium picolinate is one form of chromium III that was sold in large quantities during the “chromium craze” of the mid 1990s. It was promoted as a way to burn fat and regulate metabolism, and is still a standard ingredient in many health food supplements being sold today.34,35
As with many elements chromium can exist in several atomic valence states.
“Chromium VI (hexavalent chromium) is important to a variety of industries including pigment manufacturing and painting, metal plating, wood treating, and leather tanning…”27 Hexavalent chromium can have a toxic effect in physiological environments especially if introduced to the body in large amounts.

Chromium is a standard ingredient in many health supplements being sold today.
It is widely accepted that the valence state of chromium released from a metal-on-metal implant is primarily trivalent which is the more biocompatible form of chromium. Also, even if chromium ions were released in the hexavalent state, the levels appear to much lower than the amounts that might cause toxicity, as illustrated in the findings listed below:
• “recent kinetics and in vivo genotoxicity data demonstrate that Cr(VI) is reduced to nontoxic Cr(III) in saliva, in the acidic conditions of the stomach, and in the blood.”27
• “At concentrations at least as high as the current U.S. maximum contaminant level of 100 micrograms per liter (µg/L), and probably at least an order of magnitude higher, Cr(VI) is reduced to Cr(III) prior to, or upon systemic absorption.”27
• “Red blood cell lysates have a substantial and regenerative capacity to reduce >100 Mcg of Cr(VI) (to Cr(III) based on the hematocrit of an average adult (DeFlora and Wetterhahn, 1989; DeFlora, 1996).”29
• “Essentially all tissues possess a similar high capacity to reduce Cr(VI) to Cr(III), especially the liver, which is responsible for the “first pass effect” biotransformations of many chemicals (Sipes and Gandolfi, 1991).”29
Current metal-on-metal ion release studies have shown trivalent chromium levels in serum to be much lower than even the 100 µg/l maximum listed for hexavalent chromium.18–22 Also, most researchers agree that chromium ions released into the body from metal-on-metal implants are in the trivalent Cr(III) state, and according to the statements above, even if all of the ions occurred in the hexavalent Cr(VI) state – they could still be reduced to the trivalent state without toxic effects (Figure 6).
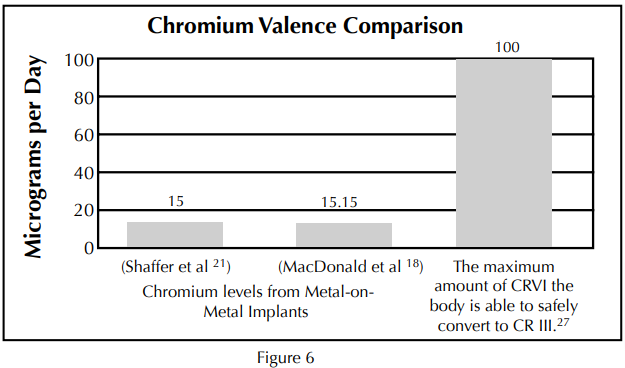
Current metal-on-metal ion release studies show trivalent chromium levels in the serum to be much lower than the 100 µg/d maximum listed for hexavalent chromium.18,21,27 Even if all of the ions occurred in the hexavalent Cr(VI) state – they could still be reduced to the trivalent state without toxic effects.
Trivalent chromium is a key element of glucose regulation and metabolic function. It has been widely consumed in pill form as a means of helping to regulate diabetes, control sugar cravings, and aid in weight loss. It has been the subject of more than 35 clinical studies involving over 2,000 participants36. The recommended daily dose as recommended by these studies is around 200 Mcg per day (Figure 7). A recent study funded by the National Institutes of Health showed that:
“Daily supplementation with 1,000 Mcg of chromium as chromium picolinate significantly enhanced insulin sensitivity. These initial results offer a potential new nutritional therapy for approximately 2 million American women suffering from Polycystic Ovarian Syndrome (PCOS). PCOS is a little-understood hormonal condition that is a leading cause of infertility, and is associated with insulin resistance, gestational diabetes and type 2 diabetes.”36 (Figure 7)
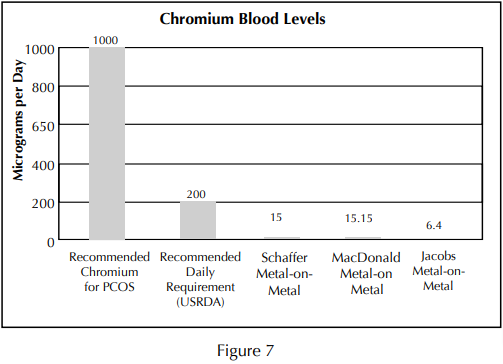
Concerns regarding the total chromium values reported in metal-on-metal ions studies should be weighed against the daily values recommend in other studies.
Cobalt also has a role in the body. Cobalt is an essential element necessary for the formation of vitamin B12, and the metabolism of proteins. Consequently, consuming extra protein and vitamin B12 (ie: meat and multivitamins), may substantially influence the urinary concentrations of cobalt. The average person consumes between 5 and 45 micrograms (mcg) of cobalt per day.30 The amount of cobalt excreted in the urine from a metal-on-metal implant appears to be between 3 and 350 Mcg per day.19-21 In the 1960s some heavy beer drinkers developed cardiac problems a couple of months after cobalt chloride was added to their local beer supply. The estimated daily intake of cobalt by these beer drinkers was 6000 to 8000 Mcg per day. Reduced protein intake and a poor diet were also cited as contributing to the beer drinker’s heart conditions30 (Figure 8). Assuming a cumulative effect of cobalt ion release from a metal-on-metal implant (in other words no release from the body) it would take approximately 438 years of cumulative cobalt ion release to compare with what the beer drinkers were ingesting in two months! Furthermore, “cancer in humans as a result of exposure to cobalt by any route has not been demonstrated.” 30 The US National Toxicology Program does not list cobalt as a recognized animal or human carcinogen.31
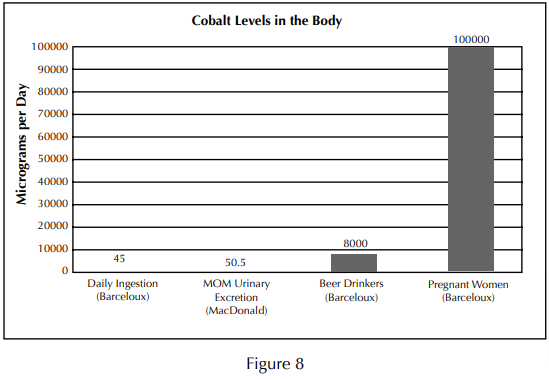
Studies of elevated cobalt levels in pregnant women, and beer drinkers have not demonstrated an elevated risk of cancer despite levels much higher than those reported from metal-on-metal implants.
5. Concerns regarding ion release must be weighed against the wear by-products of other bearing surfaces.
Every bearing surface produces an environmental burden of wear related debris, none of which are inert to the body. Occurrence of osteolysis due to excesspolythyelene wear is regularly reported in the literature.37,38 Reports of osteolysis with metalon-metal implants are rare.
Every bearing surface produces an environmental burden of wear related debris, none being totally inert in the body. Occurrence of osteolysis due to higher magnitudes of polythyelene wear has been reported in the literature.37,38 Metallic particles may also be associated with osteolysis, although reports of this are lacking, and occurrence is rare. Ceramic particles have been associated with osteolysis. Dr. Yoon, et al., reported on 103 ceramic-on-ceramic hips at 7.6 years follow-up, finding 22% femoral lysis and 49% acetabular lysis.39 Some have suggested that the highly crosslinked polyethylenes may not eliminate osteolysis, in that increased irradiation energy can lead to smaller, rounder particulate40 which has been directly associated with an increased osteolytic response.41,42 A reduced local response to metal-on-metal wear products has been well documented in twenty-year studies by Schmidt, McKellop, and Cuckler.1,2,43 Dr. Cuckler, among other researchers, has suggested that reasons for the reduced tissue response of metal-on-metal wear debris are due to ultra-low metal-on-metal wear rates which reduces the environmental burden and local inflammatory responses.43 The majority of wear products exist at a submicron level enabling the wear debris to be excreted from the body through the body’s normal metabolic processes. This was reported by Dr. Schaffer; “We conclude that the elimination of cobalt and chromium proceeds over several years, affecting a balance between release and excretion” (Figure 9). This release from the body in an ionic form may avoid a local response as well any toxic build-up on a systemic level.
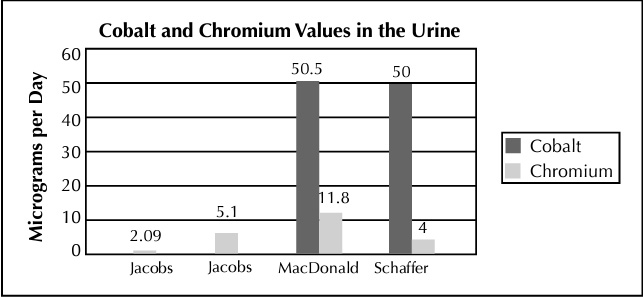
The excretion of ions through the urine is evidenced in clinical trials of metal-onmetal implants as listed above.
Conclusion
There are a variety of bearing options available for total hip replacement. Metal-on-metal is the most clinically proven alternate bearing available today, with over 40 years of clinical use. Concerns associated with metal ion release must be weighed against the risks and benefits of other bearing options. Metallic debris is an environmental by-product of metal-on-metal wear and is present in the body with non-metal-on-metal bearing surfaces.24,33 Extensive research and years of clinical trials have failed to prove any cause for concern associated with the ion levels exhibited from metal-on-metal implants. The lower-wear aspects and wide range of design options available with a metal-on-metal implant make it particularly attractive for the younger, more active patient population.

The lower-wear aspects and wide range of design options available with a metal-on-metal implant make it particularly attractive for the younger, more active patient population.
References:
- Schmidt, et al,; “Cobalt Chromium Molybdenum Metal Combination for Modular Hip Prostheses” Clinical Orthopedics and Related Research, 329, pp 35–47, August 1996.
- McKellop, et al,; “In-Vivo Wear of 3 Types of Metal on Metal Hip Prostheses During Two Decades of Use” Clinical Orthopedics and Related Research, 329, pp. 128–40, August 1996.
- Rieker, et al.; Clinical Tribological Performance of 144 Metal-on-Metal Hip Articulations. Metasul a Metal-on-Metal Bearing, Hans Huber Press, Bern, Germany; pp. 83–91, 1999.
- Jacobsson, et al,; “20-Year Results of McKee-Farrar Versus Charnley Prosthesis” Clinical Orthopedics and Related Research, 329, pp S60–S68, 1996.
- Amstutz and Grigoris,; “Metal on Metal Bearings in Hip Arthroplasty” Clinical Orthopaedics and Related Research, Number 329S, pp S11–S34, 1996.
- Lombardi, et al:, “Short-Term Results of the M2 a-Taper Metal-on-Metal Articulation” The Journal of Arthroplasty, Vol. 16(8): 122–28, December 2001.
- Dorr, et al,; “Modern Metal on Metal Articulation for Total Hip Replacements” Clinical Orthopedics and Related Research, 333, pp 108–117,1996.
- Simulator Data on File at Biomet Inc.
- Burroughs, B., Harris, W.,; “Femoral Head Size in THR: Rationale for Reassessment” Abstract 32nd Annual Advances in Hip and Knee Arthroplasty, Cambridge, MA October 2002.
- Amstutz HC., et al.,; “Range of Motion Studies for Total Hip Replacements: A Comparative Study with Experimental Apparatus”, Clinical Orthopedics and Related Research, 111, pp 124–130, 1975.
- Beaule, et al,; “Jumbo Femoral Head for the Treatment of Recurrent Dislocation Following Total Hip Replacement” The Journal of Bone and Joint Surgery, Vol. 84–A:2, pp 256–63, February 2002.
- Komistek,; R., et al., “In Vivo Comparison of Hip Separation after Metal-on-Metal or Metal-on-Polyethylene Total Hip Arthroplasty” The Journal of Bone and Joint Surgery, Volume 84A:10, pp 1836–41, October 2002.
- Elke, R., “Particle Disease: Status and Today’s Solutions” Bioceramics in Joint Arthroplasty: Proceedings of the 7th International Biolox® Symposium, March 2002.
- Li, S., “Independent Evaluation of Crosslinked Poly” Notes: 6th Annual Symposium on Alternative Bearing Surfaces in Total Joint Arthroplasty, San Francisco, CA, September 2003.
- Tharani, et al.; “The Risk of Cancer Following Total Hip or Knee Arthroplasty” The Journal of Bone and Joint Surgery, Volume 83:5, pp 774–80, May 2001.
- Visuri, et al.; “Cancer Risk After Metal on Metal and Polyethylene on Metal THA” Clinical Orthopedics and Related Research, 329, pp 280–89, August 1996.
- Merritt, et al.; “Distribution of Cobalt Chromium Wear and Corrosion Products and Biologic Reactions” Clinical Orthopedics and Related Research, 329, pp 233–43, August 1996.
- Shaffer, et al.; “Increased Blood Cobalt and Chromium After Total Hip Replacement” Clinical Toxicology, 37(7), pp 839–44, 1999.
- Brodner, et al,; “Elevated Serum Cobalt with Metal on Metal Articulating Surfaces” The Journal of Bone and Joint Surgery, Vol. 79-B:2, pp 316–21, March 1997.
- Jacobs, et al,; “Cobalt and Chromium Concentrations in Patients with Metal on Metal Total Hip Replacements” Clinical Orthopedics and Related Research, 329, pp 256–63, August 1996.
- MacDonald et al,; “Metal on Metal Versus Metal on Polyethylene Liners in THA: Clinical and Metal Ion Results of a Prospective Randomized Clinical Trial” Hip Society Abstract, February 2002.
- Skipor, A, et al., “Metal Levels in Patients with Metal-on-Metal Total Hip Replacements.” Abstract; 69th Annual Meeting Proceedings of the AAOS, Paper No. 166 Vol. 3, Dallas, TX 2002.
- Venable, C.S., and Stuck, W.G.: J.A.M.A. 111:1349, 1938.
- Kriebich, D, et al., “Systemic Release of Cobalt and Chromium After Uncemented Total Hip Replacement.” The Journal of Bone and Joint Surgery (Br) 1996;78–B:18–21.
- Maloney, WJ.: “Ceramic Bearings: America, You Arrived! – Some Cons” Session XV Paper #64 Current Concepts in Joint Replacement, May 2003.
- Aitio, A.; “Stainless Steel Kitchen Utensils as a Source of Chromium – Toxicological Implications” The Chromium File: The International Chromium Development Association Issue #1 September 1996.
- Proctor et al. Is Hexavalent Chromium Carcinogenic Via Ingestion? A Weight of Evidence of Review. Journal of Toxicology and Environmental Health, part A, 65: 701–746, 2002.
- “Toxicological Profile for Chromium” U.S. Department of Health and Human Services, pg. 13/550, September 2000.
- Kerger, et al., “Absorption and Elimination of Trivalent and Hexavalent Chromium in Humans Following Ingestion of a Bolus Dose in Drinking Water” Toxicology and Applied Pharmacology 141, 145–158 (1996).
- Barceloux, D.G. Cobalt. Clinical Toxicology, 37(2), 201–216, 1999.
- “Draft Toxicological Profile for Cobalt” U.S. Department of Health and Human Services, September 2001.
- June 2003 presentation by Dr. Wolfram Brodner at the 2nd International Conference on Metal-Metal Hip Prosthesis in Montreal
- Karamat, L. et al. Blood Analysis for Trace Metal in Metal-On-Metal and Ceramic-On-Ceramic Bearings in Total Hip Arthroplasty. (Sulzer Metasul Metal on Metal THA)
- Vincent J., “The Potential Value and Toxicity of Chromium Picolinate as a Nutritional Supplement, Weight Loss Agent and Muscle Development Agent”, Sports Medicine 2003;33(3):213–30.
- Anderson RA., “Effects of Chromium on Body Composition and Weight Loss” Nutrition Review 1998 Sep;56(9):266–70.
- 59th Annual Meeting of the American Society for Reproductive Medicine Conference (ASRM) in San Antonio, Texas) Published as a supplement to Fertility and Sterility, September 2003.
- Harris, W., “The Lysis Threshold”: An Erroneous and Perhaps Misleading Concept?” The Journal of Arthroplasty, Vol. 18 No. 4, 2003.
- Dumbleton., JH., et al., “A Literature Review of the Association Between Wear Rate and Osteolysis in Total Hip Arthroplasty” Journal of Arthroplasty, 15:649, 2002.
- Yoon, T., et al; “Osteolysis in Association with a Total Hip Arthroplasty with Ceramic Bearing Surfaces” The Journal of Bone and Joint Surgery, Vol. 80-A, No. 10, October 1998.
- Scott, M., “Wear Particle Analyses of Conventional and Crosslinked UHMWPE Tested in an Anatomic Hip Simulator”, ORS, Feb. 2001.
- Green, T., et al., “Effect of Size and Dose on Bone Resorbtion Activity of Macrophages by In Vitro Clinically Relevant UHMWPE Particles”, JBMR 53:490–497, 2000.
- Mabrey, J., et al., “Comparison of UHMWPE Particles in Synovial Fluid and Tissues from Failed THA”, JBMR 58:196–202, 2001.
- Cuckler, J.: “Results of a Prospective, Randomized Trial of Metal-Metal vs. Metal-PE THR” Abstract and Notes of The 6th Annual Alternative Bearings Symposium, San Francisco, CA, 2003.
- Head, W., et al., “Comparison of Polyethylene Wear in Machined Versus Molded Polyethylene Liners in Ringloc Acetabular Cups”, Texas Center for Joint Replacement, Plano, TX.
- Oonishi, H., et al., “Wear of High-Dose Gamma Irradiated Polyethylene in Total Joint Replacement. Long Term Radiographic Evaluation”, ORS, 97–117, 1998.
- Hamadouche M., et al., “Alumina-on-alumina total hip arthroplasty: a minimum 18.5-year follow-up study” J Bone Joint Surg Am., 2002 Jan; 84-A(1):69–77.